Contents
Overview
The genesis of rapid diagnostic tests can be traced back to early immunological assays, but their modern iteration truly began to take shape with the development of enzyme-linked immunosorbent assays (ELISAs) in the 1970s, which laid groundwork for immunoassay development. While ELISAs offered improved sensitivity, they still required laboratory infrastructure. The breakthrough for truly rapid, point-of-care diagnostics arrived with the advent of monoclonal antibodies and advancements in immunoassay technology, leading to the development of lateral flow assays. The World Health Organization (WHO) and Bill & Melinda Gates Foundation have been instrumental in driving the development and deployment of RDTs for infectious diseases in low-resource settings, particularly for malaria.
⚙️ How It Works
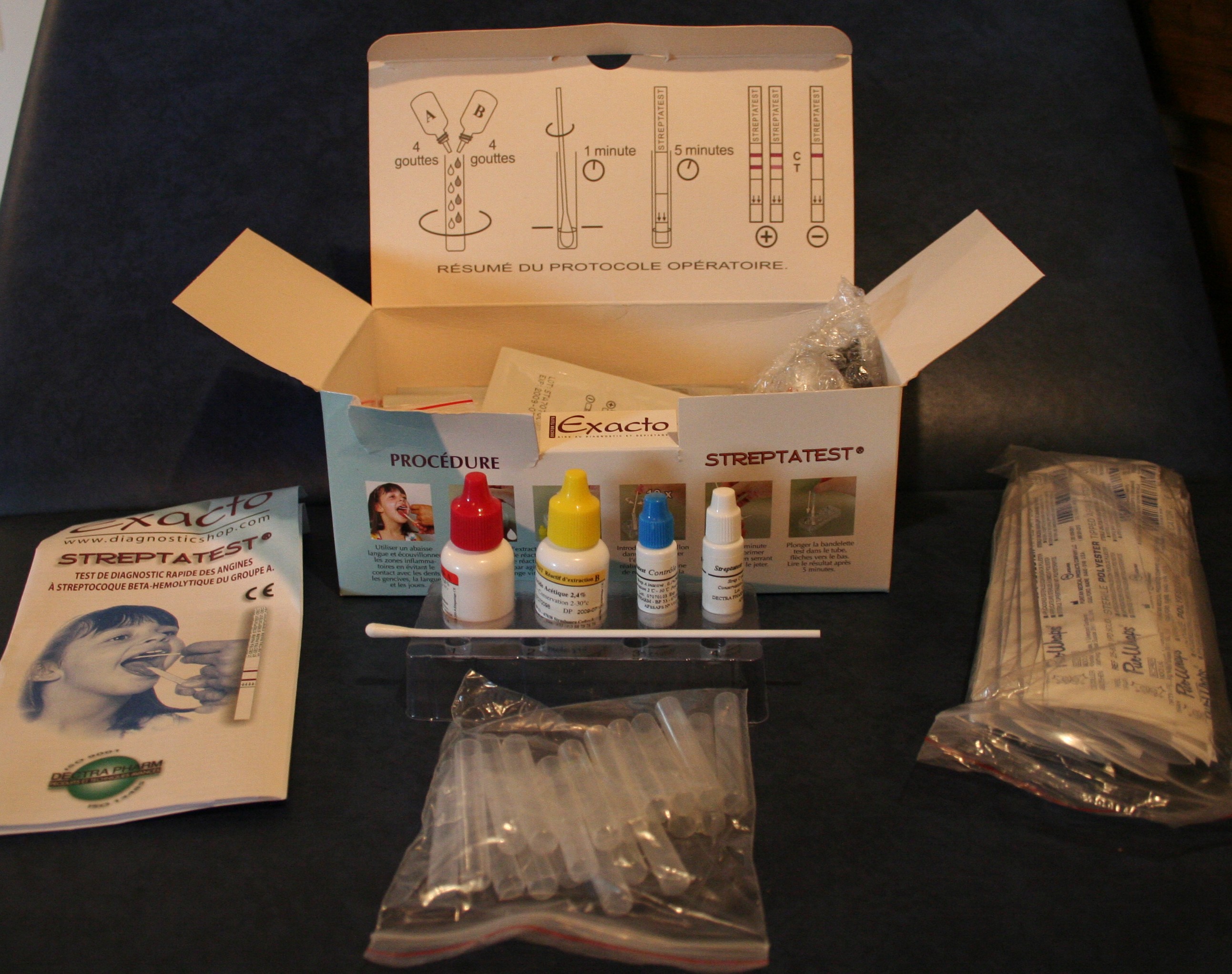
Most rapid diagnostic tests operate on the principle of immunochromatography, commonly known as lateral flow. A sample, such as blood, urine, or saliva, is applied to a sample pad. This sample then flows along a nitrocellulose membrane, encountering reagents that bind to specific target analytes (e.g., antigens or antibodies). If the target analyte is present, it forms a complex with labeled antibodies (often conjugated to colored nanoparticles like colloidal gold). This complex then migrates to a test line, where it is captured by immobilized antibodies, forming a visible line. A control line, also present on the membrane, ensures the test has run correctly. RDTs require no external power source or complex instrumentation, making them ideal for decentralized testing environments.
📊 Key Facts & Numbers
Globally, RDTs represent a significant market. During the COVID-19 pandemic, the demand for rapid antigen tests surged. RDTs account for over 60% of diagnostic testing for malaria in endemic regions, replacing microscopy in many areas. Some RDTs for infectious diseases cost less than $1 USD, a stark contrast to laboratory-based tests that can cost tens or even hundreds of dollars. Over 100 different infectious diseases can now be diagnosed using RDTs, with more than 500 different RDT products available on the market.
👥 Key People & Organizations
Key individuals and organizations have profoundly shaped the RDT landscape. The World Health Organization (WHO) plays a critical role in prequalifying RDTs for global procurement, ensuring quality and efficacy, particularly for diseases like malaria and HIV. Non-governmental organizations such as the Bill & Melinda Gates Foundation have provided substantial funding for RDT research, development, and deployment in low- and middle-income countries. Companies like Abbott Laboratories, Roche Diagnostics, and QuidelOrtho are major commercial players, developing and manufacturing a wide array of RDTs for various applications, including the widely used COVID-19 antigen tests.
🌍 Cultural Impact & Influence
Rapid diagnostic tests have democratized access to medical information, empowering individuals and healthcare providers in remote areas. The widespread availability of RDTs for diseases like malaria has dramatically improved case management in sub-Saharan Africa, reducing reliance on clinical diagnosis alone and enabling targeted treatment. During the COVID-19 pandemic, RDTs became a frontline tool for surveillance and individual screening, influencing public health policies and personal decision-making. Their ease of use has also fostered greater patient engagement in their own health, moving diagnostic capabilities from specialized laboratories directly into community health centers and even homes, fundamentally altering the patient journey.
⚡ Current State & Latest Developments
The RDT market continues to evolve rapidly, driven by technological advancements and emerging health challenges. The COVID-19 pandemic accelerated innovation, leading to more sensitive and multiplexed RDTs capable of detecting multiple pathogens simultaneously. Digital integration is a major trend, with some RDTs now incorporating smartphone connectivity to automatically record results, track data, and transmit information to public health systems, enhancing surveillance and outbreak response capabilities. Furthermore, there's a growing focus on RDTs for non-communicable diseases, such as cardiovascular markers and cancer biomarkers, expanding their utility beyond infectious disease diagnostics. CRISPR-based diagnostics also promise even greater sensitivity and specificity in the near future.
🤔 Controversies & Debates
Despite their benefits, RDTs face significant controversies. A primary concern is the variability in sensitivity and specificity across different manufacturers and test batches, leading to potential false positives or negatives. This has been particularly debated with early COVID-19 antigen tests, where performance varied widely. Regulatory oversight, while improving, can be inconsistent globally, leading to the proliferation of substandard tests. The ethical implications of deploying RDTs in resource-limited settings also arise, including ensuring adequate training for users, proper disposal of biohazardous waste, and equitable access to follow-up treatment based on test results. The debate over whether RDTs should replace, supplement, or be used in conjunction with laboratory-based diagnostics remains ongoing for many conditions.
🔮 Future Outlook & Predictions
The future of RDTs points towards greater integration with digital health platforms and enhanced analytical capabilities. Expect to see more multiplexed tests capable of detecting a panel of infectious agents or biomarkers simultaneously, offering a more comprehensive diagnostic picture from a single sample. The integration of AI and machine learning with RDT data will enable more sophisticated analysis, predictive diagnostics, and personalized treatment recommendations. Advancements in nanotechnology and biotechnology, such as CRISPR-based detection systems, promise RDTs with laboratory-grade sensitivity and specificity, potentially revolutionizing diagnostics for everything from early cancer detection to antibiotic resistance monitoring. The goal is to make highly accurate diagnostics accessible anywhere, anytime.
💡 Practical Applications
Rapid diagnostic tests have a vast array of practical applications across healthcare and public health. They are indispensable for diagnosing infectious diseases like malaria, HIV, influenza, and COVID-19 at the point of care, enabling immediate treatment and reducing disease transmission. In primary care, RDTs are used for strep throat detection, urinalysis, and monitoring chronic conditions like diabetes (e.g., blood glucose monitoring). They are critical in emergency medicine for rapid assessment of conditions such as cardiac markers (e.g., troponin) or drug screening. Furthermore, RDTs are deployed in outbreak surveillance, mass screening campaigns, and in remote or austere environments where laboratory access is limited, such as military field operations or disaster relief efforts.
Key Facts
- Category
- technology
- Type
- topic